In 2021, the equivalent of 1.7 planets has been used to satisfy our production and consumption needs1. Faced with this type of alarming observation, CSR issues are fortunately carrying more and more weight in companies. Thus, many industrial sectors are reconsidering their methods in sourcing, production, or transport to develop more sustainable and sounder ecosystems.
Several approaches are available, and biotechnology has been identified as one of the major levers to meet the challenges of a production that combines both environmental protection and economic performance. Indeed, biotechnologies are increasingly attractive and accessible in the industrial sector. This is even more true in the field of white biotechnologies, which are dedicated to the alternative and bio-based production of chemical, material, and energy compounds.
Fermentation plays a key role in white biotechnology since it allows for the production of the molecules that will be used in industrial applications. But what is the precise role of fermentation in the development of the bioeconomy? What are the associated issues? Find out what you need to know about industrial fermentation in this article!
Principles of fermentation: an historical and advanced process
How does fermentation work?
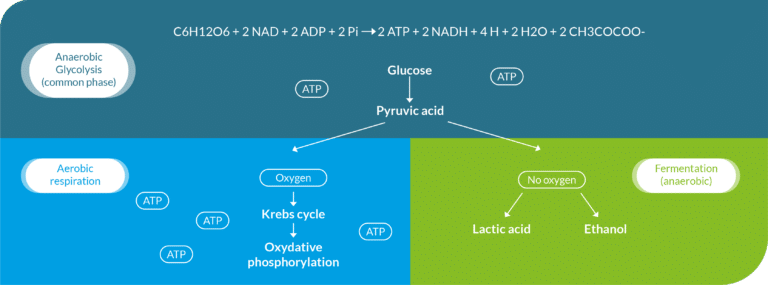
Before getting into the subject matter, let’s do a quick but essential theoretical review. Fermentation is a biological process that occurs in either an oxygen-free environment –under anaerobic conditions – or under aerobic conditions – with the presence oxygen. It is the transformation of an organic material by the enzymes that are released from bacterias, yeasts or molds present in the environment. These microorganisms feed on properties of material, especially glucose and water. They are charged with energy (ATP), reproduce, and then produce acids, gases or alcohol that can be of interest to humans –making food more digestible or preserving it longer, for instance2.
Each of these microorganisms has specific fermentation capacities that lead to different types of fermentation – ethyl, lactic, butyric, sulfuric, malolactic – whose released substrates can be used in different ways.
Fermentation has been used by man since its sedentarization between 8,000 and 4,000 BC and its increasing mastery has allowed it to expand its uses.
Industrial fermentation: multiple applications
The notion of biotechnologies appeared in the 20th century, with the beginnings of enzymology and Pasteur’s work on fermentation. From then on, microorganisms were used to create various products of interest such as vaccines, antibiotics, or baker’s yeast. Many business sectors thus benefit from the advantages of fermentation.
Many uses can be named for the food industry alone: bread making, taste improvement, reproduction of flavors or creation of fermented beverages, to mention just a few. Fermentation is also widely used in the health sector, whether for the supply of preventive or curative nutrients, the development of pharmaceutical products or to encourage a healthier agriculture for example.
On the industrial side, one of the most common applications is the production of bioethanol, with more than 27 million gallons produced per year worldwide. However, industrial players do not always know that microorganisms can be integrated into many other manufacturing processes as a replacement for petroleum products.
A few examples: methanation is used for renewable energy production and waste treatment, bio-based fibers are used to obtain biodegradable textiles and clothing, bio-based molecules are used to create substitutes for certain materials, such as plastic. And the examples of industrial biotechnology projects that use fermentation are more and more numerous!
Fermentation as a way to reach a sustainable industry
Fermentation: an eco-friendly process
Advantages of fermentation are numerous, and the environment is the first beneficiary. The development of industrial biotechnology, especially through fermentation processes, fully participates in the establishment of a virtuous and widespread bioeconomy. The use of bio-based products can indeed reduce the environmental and ecological impact of the industry, in particular by reducing its CO2 emissions, for many reasons:
- It is an alternative to the use of fossil fuels. Petrochemical molecules can be totally or partially substituted by bio-based molecules, while providing an identical or even improved final material.
- The efficacy of a fermentation process sometimes allows to consume less energy and water.
- Products made from renewable or bio-based materials are toxic-chemicals-free: biodegradable plastics, biopesticides, organic and biodegradable household products…
- Bio-based molecules can replace animal-derived materials, particularly in cosmetics.
- Bio-based productions generate less toxic waste.
Although the positive impact of fermentation on the environment is proven, major progress remains to be done. Less than 15% of Europe’s renewable energy comes from bio-based materials. According to European Bioplastics, in 2018, biobased plastics accounted for only 1% of global 335 million tons plastic production.
Bio-based chemistry, on the other hand, currently represents only a very small share of all organic chemistry: one to two million tons out of approximately 400 million tons produced per year3. However, let’s stay on a positive note: it is estimated that it should reach 15 million tons by 2030.
Industrial fermentation as an efficient and profitable tool
The benefits of fermentation are obviously not limited to environmental aspects. The development of biotechnologies, combined with the mastery of fermentation processes and the sequencing of microorganism genomes, also provides numerous advantages in terms of performance.
The process of selection, optimization and development of strains allows the elaboration of very robust industrial strains that are able to withstand the specific conditions of industrial processes and to meet precise objectives. The resulting products can be improved versions of existing ones or completely new products, with highly sought-after functionalities or properties, such as resistance or lightness. They can be produced in different physiological states as required, genetically modified or not, to achieve the desired capacities.
The advantages of integrating bio-based molecules into industrial processes can then be numerous:
- Improvement of process efficiency
- Development of new product features
- Reduction of the quantity of needed raw materials
- Reduction of the volume of produced waste
- Reduction of toxicity both of the process and the final product
- Reduction of production costs
- Reduction of the environmental impact of the activity
- Additional element for competitor differentiation
- Improvement of supply reliability
- Reduction of transportation costs (in case of local sourcing)
However, performance-related arguments are not always enough to convince manufacturers to take the plunge. Economic profitability is of course an absolute priority, and the adoption of bio-based processes is therefore very dependent on the return on investment. It is only attractive if its costs are similar or lower than those of products from the petrochemical industry. This opportunity relative to the cost obviously differs according to the projects and remains fluctuating, since it is entirely dependent on the prices of raw materials, in particular the price of oil. The cost of adoption also depends on the material investments to be made.
On the other hand, the current context of rising energy prices is clearly conducive to the development of the bioeconomy, as could be some regulations that are still to come. Fermentation is therefore a key process, which offers infinite possibilities for innovation, and which is already, or could become, highly profitable.
Scale-up: the key challenge of industrial fermentation
From lab-scale to industrial-scale fermentation: a big step forward
Numerous fermentation processes have been developed in the laboratory. But the challenge of industrial fermentation lies in its scaling up, both technically and economically. The conditions of culture are indeed completely different for a high volume production and in an industrial context. Exact duplicates of the pilot production will not lead to the same quality and performance. Even the choice of microorganism used at the beginning may not be suitable for scale-up.
The process and the final product are intimately linked in fermentation, unlike in classical industrial processes development. This specificity must be taken into account in the search for an optimal industrial production, with a precise experimental planning and a meticulous analytical follow-up of all the fermentation stresses having an influence on the sterility, quality, regularity, and performance of the production: raw material, pressure, pH, temperature, agitation, contamination, cell ageing, etc…
Success of scaling up is therefore conditioned by the use of the right development tools. For example, our internal specific innovation platform, CP², addresses these challenges in the most reliable and optimal way.
As with any biochemical process, fermentation performance can be assessed through productivity, yield and titer indicators, which directly impact the plant’s results and the company’s profitability. A strong issue of economic viability thus lies behind the technical challenge.
Making your scale-up a success
While successfully scaling up fermentation processes is a real challenge, companies have no less responsibility to redefine the economy by moving towards more sustainable systems, whether by conviction or obligation.
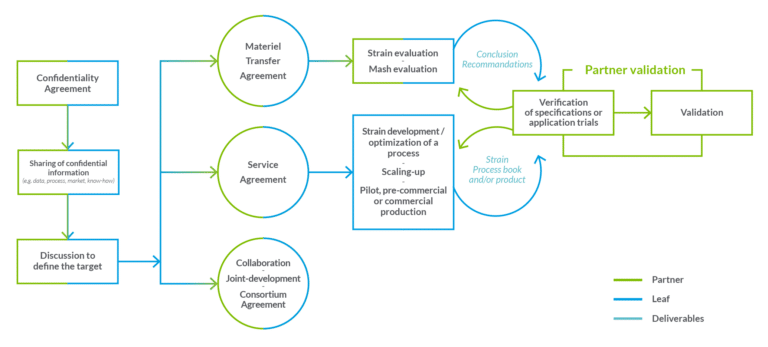
To be accompanied by experts is a way to develop, and especially to succeed in the use of fermentation within production processes. Players like Leaf provide their skills and know-how and intervene by the industrialists at each stage of the fermentation process:
- Strain selection and engineering: selection from a relevant and diverse microorganisms collection and development of industrial strains with high robustness to meet needs.
- Optimization of fermentation processes to produce biomass and/or molecules that mimic industrial conditions.
- Scale-up: increase fermentation volumes within specific facilities and equipment, under industrial or similar conditions, to test the economic viability of the developed process. Once the process and quality are validated, samples are provided for further testing to prove the suitability of the solution at industrial scale.
- From pilot to commercial production: the commercial production stage can also be integrated into the process, with the existence of contract fermentation. This allows the production of proprietary GMO and non-GMO microorganisms on a commercial scale, thus ensuring on-time supply. In the case of contract fermentation, working in compliance with the applicable regulations, as well as with compliant partners, is at the heart of our collaboration process.
Building partnerships
There are multiple benefits in partnering when it comes to industrial fermentation:
- Benefiting from recognized industrial expertise, taking into account the critical scales and key factors necessary for industrialization
- De-risking innovation by demonstrating expected performance through feasibility studies and pre-evaluations.
- Benefiting from a continuous improvement service, with ongoing R&D activities feeding existing and new partners’ production processes.
- Addressing quality issues, thanks to a contractualized quality guarantee and a proven process development method
- Benefiting from supply reliability, with the existence of back-up production sites and contractual guarantees.
- Benefiting from a complete technical support: team training, data exploitation and analysis, parameterization, microbiology test , troubleshooting…






